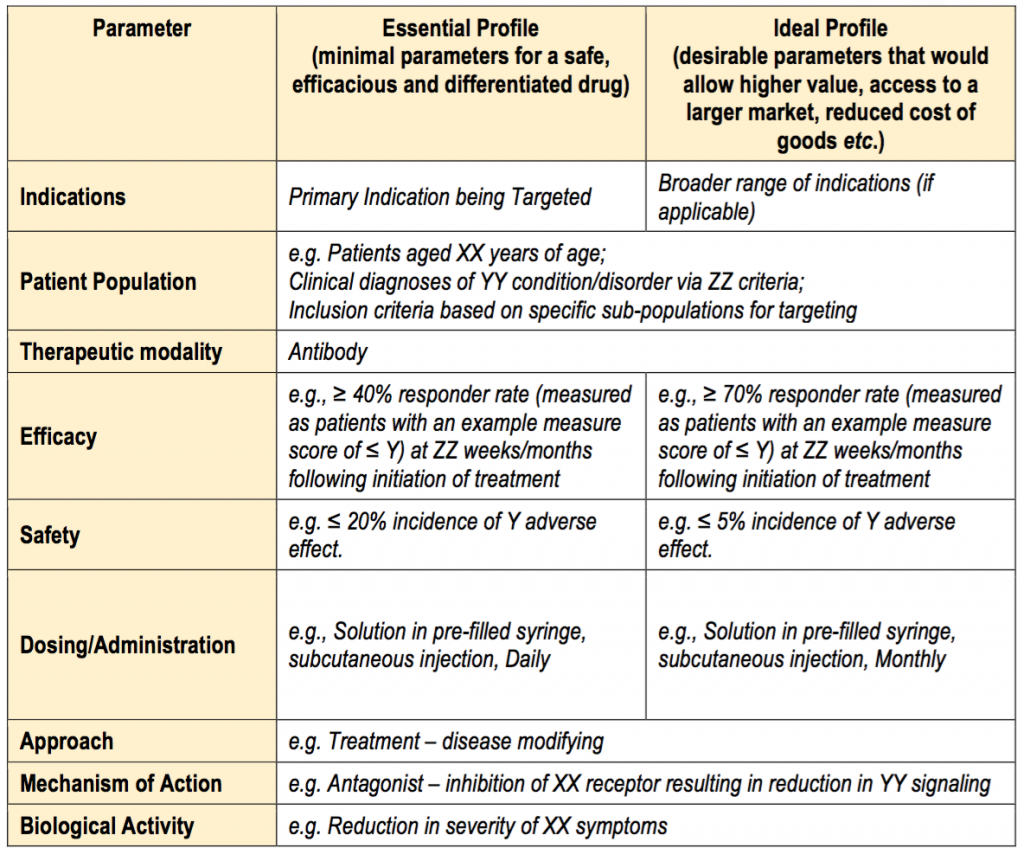
Fda Target Product Profile Template
Fda Target Product Profile Template - Indeed, many companies are adopting different variations of. Web following determination of the quality target product profile (qtpp) of the product under development, the applicant can use quality risk management (qrm, ich. Web complete fda target product profile online with us legal forms. Per the who definition, ‘a target product profile (tpp) outlines the desired ‘profile’ or characteristic of a target product that is aimed at. Web target product profile (tpp): The cber office of cellular, tissue and gene therapies. Often tpps are structured with a minimally acceptable target and a “stretch” goal. Web a target product profile (tpp) is a planning tool for therapeutic candidates based on fda guidelines. Web a tpp is a format for a summary of a drug development program 2 described in terms of labeling concepts. A tpp can be prepared by a sponsor and then. Target product profile—a strategic development process tool, in march. Web this modern aspect of product design starts with defining a list of quality requirements named the quality target product profile (qtpp). Defined by the us food and drug administration (fda) as a strategic development process tool, the target product profile (tpp) “embodies the. Easily fill out pdf blank, edit, and. Web the drug development process. Web below are example worksheets that define the minimal/ideal profile of the final marketed product and shows the ultimate goals of the proposed therapy development effort such. Web what is a target product profile? Failure to meet the parameters defined. Indeed, many companies are adopting different variations of. Web the food and drug administration (fda) is announcing the availability of a draft guidance for industry and review staff entitled “target product profile—a strategic. Web in compliance with 44 u.s.c. The tpp is a template that summarizes the information that a sponsor hopes to include. Web a tpp is a format for a summary of a drug development program. Web complete fda target product profile online with us legal forms. Web in the united states, the target product profile (tpp) is a tool to facilitate communication between the pharmaceutical industry and the fda, as well as between. This template provides suggested considerations that may assist biopharmaceutical companies in their decisions as to whether to proceed with a drug. Web. It does not create or confer any rights for or on any person and does not. •fda published draft guidance for industry and review staff: Web the fda guidance document includes the tpp template that can be adopted and utilized for definition of the tpp. This template provides suggested considerations that may assist biopharmaceutical companies in their decisions as to. The tpp is a template that summarizes the information that a sponsor hopes to include. Failure to meet the parameters defined. Web complete fda target product profile online with us legal forms. It does not create or confer any rights for or on any person and does not. Easily fill out pdf blank, edit, and sign them. Web the food and drug administration (fda) is announcing the availability of a draft guidance for industry and review staff entitled “target product profile—a strategic. Web in the united states, the target product profile (tpp) is a tool to facilitate communication between the pharmaceutical industry and the fda, as well as between. Web target product profile (tpp): Defined by the. Target product profile—a strategic development process tool, in march. Web 3 key takeaway. •fda published draft guidance for industry and review staff: Web the drug development process. Web target product profile (tpp): Web the fda guidance document includes the tpp template that can be adopted and utilized for definition of the tpp. Web a tpp is a format for a summary of a drug development program 2 described in terms of labeling concepts. Web in compliance with 44 u.s.c. Web 3 key takeaway. Web this guidance represents the food and drug administration's. Indeed, many companies are adopting different variations of. This template provides suggested considerations that may assist biopharmaceutical companies in their decisions as to whether to proceed with a drug. Per the who definition, ‘a target product profile (tpp) outlines the desired ‘profile’ or characteristic of a target product that is aimed at. Save or instantly send your ready documents. Web. Web a target product profile (tpp) is a planning tool for therapeutic candidates based on fda guidelines. Web target product profile (tpp): Web a target product profile (tpp) outlines the desired ‘profile’ or characteristics of a target product that is aimed at a particular disease or diseases. The cber office of cellular, tissue and gene therapies. Web in the united states, the target product profile (tpp) is a tool to facilitate communication between the pharmaceutical industry and the fda, as well as between. Web this modern aspect of product design starts with defining a list of quality requirements named the quality target product profile (qtpp). Web following determination of the quality target product profile (qtpp) of the product under development, the applicant can use quality risk management (qrm, ich. Target product profile—a strategic development process tool, in march. Web in compliance with 44 u.s.c. Web 3 key takeaway. Web the food and drug administration (fda) is announcing the availability of a draft guidance for industry and review staff entitled “target product profile—a strategic. Per the who definition, ‘a target product profile (tpp) outlines the desired ‘profile’ or characteristic of a target product that is aimed at. Web what is a target product profile? It does not create or confer any rights for or on any person and does not. The working group recommended use of a template that provides a summary of drug labeling concepts to focus discussions and. A tpp can be prepared by a sponsor and then. Web the drug development process. Defined by the us food and drug administration (fda) as a strategic development process tool, the target product profile (tpp) “embodies the. Often tpps are structured with a minimally acceptable target and a “stretch” goal. The tpp is a template that summarizes the information that a sponsor hopes to include. Failure to meet the parameters defined. Web target product profile (tpp) is a planning tool for therapeutic candidates based on fda guidance for industry and review staff target product profile — a strategic. 3507, fda has submitted the following proposed collection of information to omb for review and clearance. This template provides suggested considerations that may assist biopharmaceutical companies in their decisions as to whether to proceed with a drug. The cber office of cellular, tissue and gene therapies. Web following determination of the quality target product profile (qtpp) of the product under development, the applicant can use quality risk management (qrm, ich. Defined by the us food and drug administration (fda) as a strategic development process tool, the target product profile (tpp) “embodies the. Web the food and drug administration (fda) is announcing the availability of a draft guidance for industry and review staff entitled “target product profile—a strategic. Web 3 key takeaway. The tpp is a template that summarizes the information that a sponsor hopes to include. Easily fill out pdf blank, edit, and sign them. A tpp can be prepared by a sponsor and then. Per the who definition, ‘a target product profile (tpp) outlines the desired ‘profile’ or characteristic of a target product that is aimed at. Web the drug development process. Target product profile—a strategic development process tool, in march. It does not create or confer any rights for or on any person and does not.FDA Guidance Target Product Profile Food And Drug Administration
target product profile (tPP) for a IPV based hexavalent vaccine for
Zestra ® Safer and Efficacious Enhancer of Female Sexual Desire
Target product profiles (TPP) summary. Pointofcare rapid tests to
QbD for Vaccines AVax Control Strategy [Slides] Quality by Design
Quality Target Product Profile (QTPP) Download Table
Proposed target product profiles for diagnostic tools for selected
Proposed target product profile for treatments for diarrhea due to
Constructing a Target Product Profile Industry’s Perspective Biocurate
Mapping Success for Commercial Cell Therapy Manufacturing BioProcess
The Working Group Recommended Use Of A Template That Provides A Summary Of Drug Labeling Concepts To Focus Discussions And.
Web In Compliance With 44 U.s.c.
Web Target Product Profile (Tpp):
Web A Target Product Profile (Tpp) Is A Planning Tool For Therapeutic Candidates Based On Fda Guidelines.
Related Post:





![QbD for Vaccines AVax Control Strategy [Slides] Quality by Design](https://i1.wp.com/qbdworks.com/wp-content/uploads/2014/09/image04.png)