Transcelerate Protocol Template
Transcelerate Protocol Template - Plus, resources to customer their use,. As illustrated, clinical trial results can be simulated to illustrate the impact of the different options for post randomisation events to stakeholders. Plus, related to support their use, implementation, and. The cpt is a harmonized, streamlined approach to formatting and adding content to clinical trial protocols, and is intended for. Web the transcelerate protocol template approach now includes the estimand attributes, which has resulted in greater transparency and clarity regarding study objectives. Web this video explores out common protocol template (cpt) initiative and highlights how a harmonized cpt can simplify trial protocols, regulatory reviews and benefit multiple. Web transcelerate’s common protocol template (cpt) is a protocol template with common elements and structure, suitable for adoption across the industry. Resources for our new common protocol template is now accessible via download. The transcelerate common protocol template initiative represents the first time when a core group of 18 biopharma companies came together and agreed. Today announced the availability of an enhanced technology enabled common protocol. As illustrated, clinical trial results can be simulated to illustrate the impact of the different options for post randomisation events to stakeholders. Web a template to assist in the identification of documentation of protocol specific “important deviations.” to download the excel pdap template please click here. Web july 8, 2019 | growing numbers of study sponsors have been adopting the. Web a template to assist in the identification of documentation of protocol specific “important deviations.” to download the excel pdap template please click here. Plus, related to support their use, implementation, and. Web templates for the common protocol (cpt), stats analysis plan (sap), and clinical study report (csr) are available here. The cpt is a harmonized, streamlined approach to formatting. Web a template to assist in the identification of documentation of protocol specific “important deviations.” to download the excel pdap template please click here. Web i am happy to share tangible proof that the clear answer is “yes!”. Web the transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and. Today, nih and fda released the final version of a template document i blogged about last march. Today announced the availability of an enhanced technology enabled common protocol. Web the transcelerate protocol template approach now includes the estimand attributes, which has resulted in greater transparency and clarity regarding study objectives. Web templates for the common protocol (cpt), statistical analysis schedule. Plus, related to support their use, implementation, and. Web the transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration. Web january 21, 2016. Web of the publicly available templates, only transcelerate’s common protocol template (cpt) addresses the estimands framework. Web the transcelerate protocol template approach now includes. Web the transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration. The transcelerate common protocol template initiative represents the first time when a core group of 18 biopharma companies came together and agreed. As illustrated, clinical trial results can be simulated to illustrate the impact of the. Plus, related to support their use, implementation, and. Web i am happy to share tangible proof that the clear answer is “yes!”. Web transcelerate’s common protocol template (cpt) is a protocol template with common elements and structure, suitable for adoption across the industry. Web templates for the common protocol (cpt), statistical analysis schedule (sap), also clinical study view (csr) are. Web january 21, 2016. Web templates for the common protocol (cpt), statistical analysis schedule (sap), also clinical study view (csr) are available here. Web as transcelerate’s csr template represents an important milestone in authoring csrs, we offer csr authors advice and recommendations on its use,. Resources for our new common protocol template is now accessible via download. Plus, related to. The transcelerate common protocol template initiative represents the first time when a core group of 18 biopharma companies came together and agreed. Web january 21, 2016. Web a template to assist in the identification of documentation of protocol specific “important deviations.” to download the excel pdap template please click here. The cpt is a harmonized, streamlined approach to formatting and. Web the transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration. Web january 21, 2016. Web templates for the common protocol (cpt), stats analysis plan (sap), and clinical study report (csr) are available here. Web the transcelerate protocol template approach now includes the estimand attributes, which has. Plus, related to support their use, implementation, and. Web january 21, 2016. As illustrated, clinical trial results can be simulated to illustrate the impact of the different options for post randomisation events to stakeholders. Web transcelerate’s common protocol template (cpt) is a protocol template with common elements and structure, suitable for adoption across the industry. Web i am happy to share tangible proof that the clear answer is “yes!”. Web templates for the common protocol (cpt), statistical analysis schedule (sap), also clinical study view (csr) are available here. Web templates for the common protocol (cpt), stats analysis plan (sap), and clinical study report (csr) are available here. The cpt is a harmonized, streamlined approach to formatting and adding content to clinical trial protocols, and is intended for. Web the transcelerate protocol template approach now includes the estimand attributes, which has resulted in greater transparency and clarity regarding study objectives. Web of the publicly available templates, only transcelerate’s common protocol template (cpt) addresses the estimands framework. Web as transcelerate’s csr template represents an important milestone in authoring csrs, we offer csr authors advice and recommendations on its use,. The transcelerate common protocol template initiative represents the first time when a core group of 18 biopharma companies came together and agreed. Web a template to assist in the identification of documentation of protocol specific “important deviations.” to download the excel pdap template please click here. Web the transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration. Today announced the availability of an enhanced technology enabled common protocol. Web this video explores out common protocol template (cpt) initiative and highlights how a harmonized cpt can simplify trial protocols, regulatory reviews and benefit multiple. Today, nih and fda released the final version of a template document i blogged about last march. Resources for our new common protocol template is now accessible via download. Plus, resources to customer their use,. Web july 8, 2019 | growing numbers of study sponsors have been adopting the common protocol template (cpt)—or at least its basic structure—since transcelerate biopharma released the first version in 2015. Web of the publicly available templates, only transcelerate’s common protocol template (cpt) addresses the estimands framework. Web transcelerate’s common protocol template (cpt) is a protocol template with common elements and structure, suitable for adoption across the industry. Web this video explores out common protocol template (cpt) initiative and highlights how a harmonized cpt can simplify trial protocols, regulatory reviews and benefit multiple. Web the transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration. Web january 21, 2016. Web the transcelerate protocol template approach now includes the estimand attributes, which has resulted in greater transparency and clarity regarding study objectives. Web templates for the common protocol (cpt), statistical analysis schedule (sap), also clinical study view (csr) are available here. Web july 8, 2019 | growing numbers of study sponsors have been adopting the common protocol template (cpt)—or at least its basic structure—since transcelerate biopharma released the first version in 2015. The cpt is a harmonized, streamlined approach to formatting and adding content to clinical trial protocols, and is intended for. Web templates for the common protocol (cpt), stats analysis plan (sap), and clinical study report (csr) are available here. The transcelerate common protocol template initiative represents the first time when a core group of 18 biopharma companies came together and agreed. Plus, resources to customer their use,. Resources for our new common protocol template is now accessible via download. Today announced the availability of an enhanced technology enabled common protocol. Web a template to assist in the identification of documentation of protocol specific “important deviations.” to download the excel pdap template please click here. Plus, related to support their use, implementation, and.TransCelerate Common Technical Protocol integration into Starting Point
Clinical Trial Protocol Template Ema
Protocol Template Improve Your Business Easily and Fast
TransCelerateCurriculumVitaeForm.pdf Clinical Trial Health Sciences
Research Protocol Template
Protocol template transcelerate CONFIDENTIAL Protocol protocol
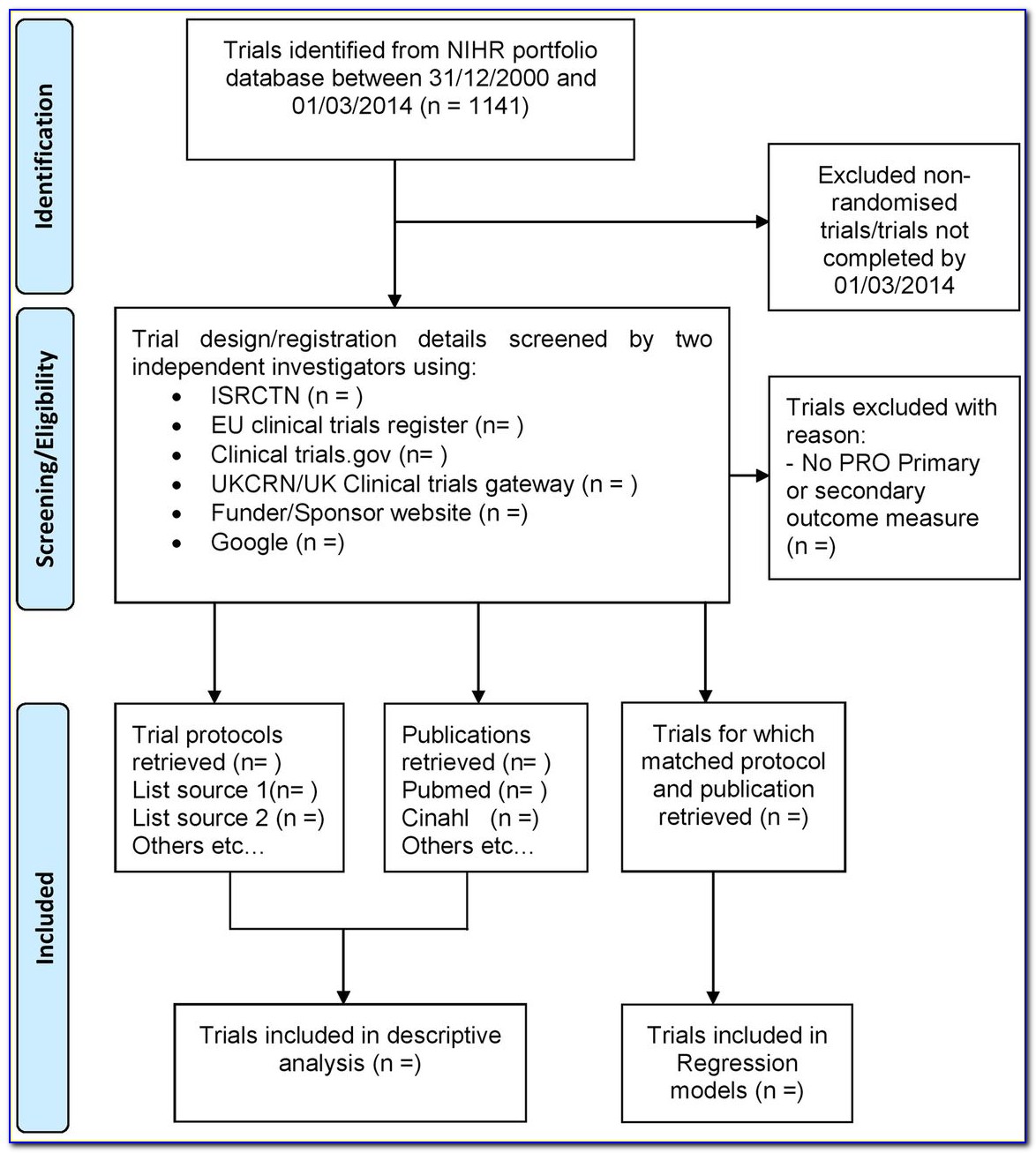
(PDF) Critical review of the TransCelerate Template for clinical study
Protocol Template Word My XXX Hot Girl
(PDF) Efficacy of combination therapy with ezetimibe and statins versus
Study Protocol Template Gambaran
As Illustrated, Clinical Trial Results Can Be Simulated To Illustrate The Impact Of The Different Options For Post Randomisation Events To Stakeholders.
Web As Transcelerate’s Csr Template Represents An Important Milestone In Authoring Csrs, We Offer Csr Authors Advice And Recommendations On Its Use,.
Today, Nih And Fda Released The Final Version Of A Template Document I Blogged About Last March.
Web I Am Happy To Share Tangible Proof That The Clear Answer Is “Yes!”.
Related Post: