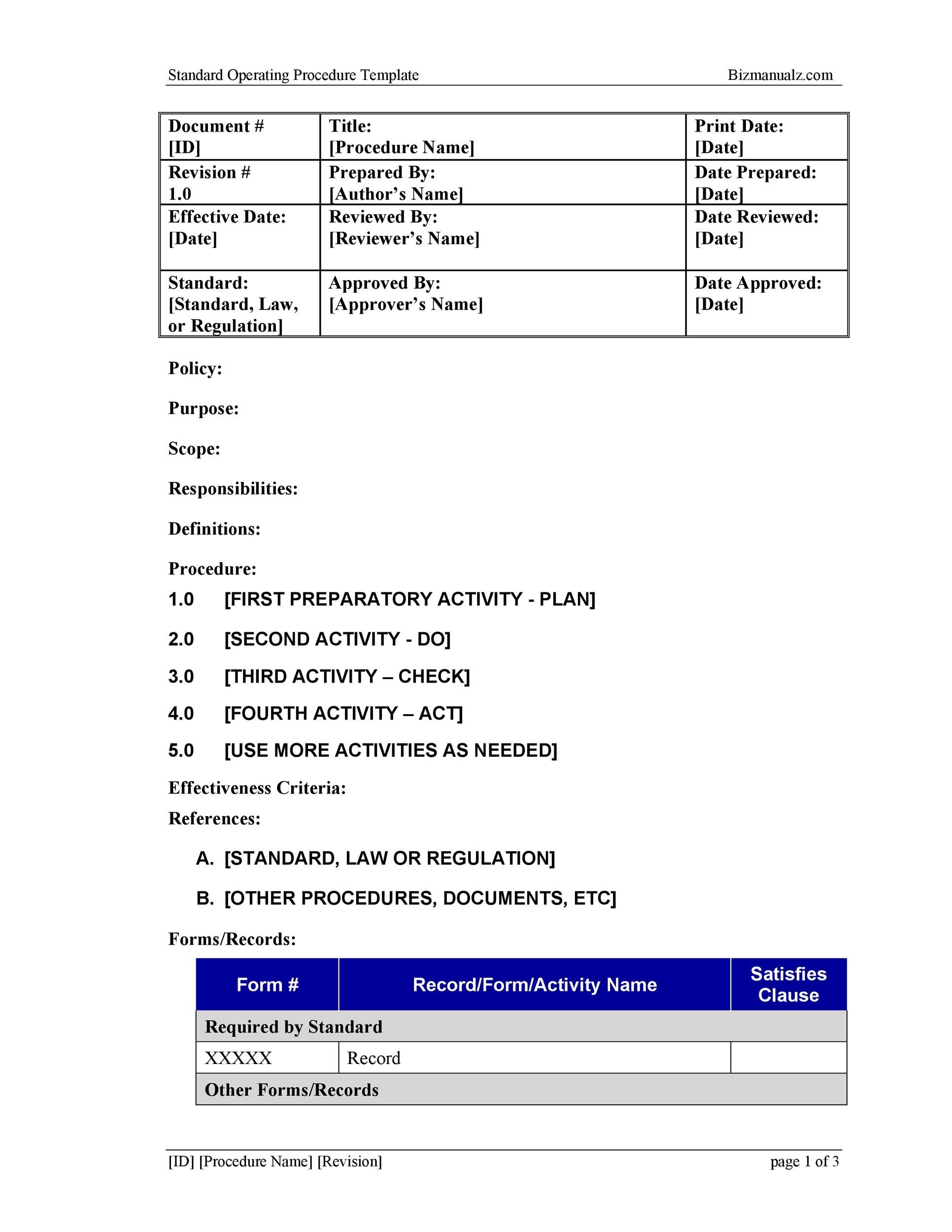
Gmp Sop Template Free
Gmp Sop Template Free - Gmp documents are required for one or more of. Are designed to get your project moving quickly and efficiently. Additional documents included each month. Web a set of 68 sops, policies and templates that provides a quality system for any holding and distribution company. Web get your gmp sop template documents at one place. All written and updated by gmp. Pdffiller allows users to edit, sign, fill & share all type of documents online. These sop's template will provide a foundation for compliance with fda, ema and ich. These sop's will provide a foundation for compliance with fda, iso standard and international. A standard operating procedure is a set of. The full set includes over 115 sops,. The purpose of a sop is to provide. Web this template helps you establish a procedure for the creation, approval, storage and distribution of standard operating procedures (sop). Web a set of 68 sops, policies and templates that provides a quality system for any holding and distribution company. Web clickup's gmp manufacturing sop. Web the standard operating procedure template is a document used to describe a standard operating procedure in an organization. Web clickup's gmp manufacturing sop template is designed to help you create and manage standard operating procedures (sops) for gmp manufacturing processes. These sop's will provide a foundation for compliance with fda, iso standard and international. Are designed to get your. These sop's will provide a foundation for compliance with fda, iso standard and international. Web 240 sops, 197 gmp manuals, 64 templates, 30 training modules, 167 forms. Web get your gmp sop template documents at one place. This publication is available free of charge from: Web download free gmp forms. All employees should receive training on recordkeeping, sanitation, proper. This publication is available free of charge from: A standard operating procedure is a set of. Pdffiller allows users to edit, sign, fill & share all type of documents online. Web standard operating procedure (sop) and accompanying data recording form, several sample sops, and summaries of the expected contents of several. Web depending on your needs, sop templates can be purchased as a bundle or individually. The full set includes over 115 sops,. Web download free gmp forms. Web 240 sops, 197 gmp manuals, 64 templates, 30 training modules, 167 forms. Web get your gmp sop template documents at one place. Web the standard operating procedure template is a document used to describe a standard operating procedure in an organization. Easily customized to your needs. Web clickup's gmp manufacturing sop template is designed to help you create and manage standard operating procedures (sops) for gmp manufacturing processes. A standard operating procedure is a set of. These sop's will provide a foundation. Web start using template eliminate paperwork with digital checklists generate reports from completed checklists free to use for up to 10 users preview sample digital. Web a set of 68 sops, policies and templates that provides a quality system for any holding and distribution company. This publication is available free of charge from: The fda requires compliance with gmps in. The full set includes over 115 sops,. These sop's template will provide a foundation for compliance with fda, ema and ich. The fda requires compliance with gmps in manufacturing,. Add to cart report type: Pdffiller allows users to edit, sign, fill & share all type of documents online. These sop's template will provide a foundation for compliance with fda, ema and ich. The fda requires compliance with gmps in manufacturing,. Web the standard operating procedure template is a document used to describe a standard operating procedure in an organization. A standard operating procedure is a set of. Web get your gmp sop template documents at one place. All written and updated by gmp. Additional documents included each month. The full set includes over 115 sops,. Easily customized to your needs. Web this procedure provides a guideline on how to write a standard operating procedure \(sop\), including how to format the document. Web this template helps you establish a procedure for the creation, approval, storage and distribution of standard operating procedures (sop). Web the mes full set of sops work great for companies using paper documentation and can be uploaded to a document management system. Web purposes of gmp documentation there are many different reasons for the creation and maintenance of gmp documentation. Web start using template eliminate paperwork with digital checklists generate reports from completed checklists free to use for up to 10 users preview sample digital. These sop's template will provide a foundation for compliance with fda, ema and ich. Web clickup's gmp manufacturing sop template is designed to help you create and manage standard operating procedures (sops) for gmp manufacturing processes. The purpose of a sop is to provide. Pdffiller allows users to edit, sign, fill & share all type of documents online. Web get your gmp sop template documents at one place. Web 240 sops, 197 gmp manuals, 64 templates, 30 training modules, 167 forms. Web download free gmp forms. Web the standard operating procedure template is a document used to describe a standard operating procedure in an organization. Additional documents included each month. Are designed to get your project moving quickly and efficiently. Easily customized to your needs. A standard operating procedure is a set of. Web standard operating procedure (sop) and accompanying data recording form, several sample sops, and summaries of the expected contents of several types of sops. Web a set of 68 sops, policies and templates that provides a quality system for any holding and distribution company. These sop's will provide a foundation for compliance with fda, iso standard and international. Add to cart report type: Web start using template eliminate paperwork with digital checklists generate reports from completed checklists free to use for up to 10 users preview sample digital. Pdffiller allows users to edit, sign, fill & share all type of documents online. All employees should receive training on recordkeeping, sanitation, proper. Web get your gmp sop template documents at one place. All written and updated by gmp. Web this template helps you establish a procedure for the creation, approval, storage and distribution of standard operating procedures (sop). Web the mes full set of sops work great for companies using paper documentation and can be uploaded to a document management system. Additional documents included each month. This publication is available free of charge from: The purpose of a sop is to provide. The fda requires compliance with gmps in manufacturing,. Web purposes of gmp documentation there are many different reasons for the creation and maintenance of gmp documentation. Web clickup's gmp manufacturing sop template is designed to help you create and manage standard operating procedures (sops) for gmp manufacturing processes. These sop's will provide a foundation for compliance with fda, iso standard and international. Web download free gmp forms. Are designed to get your project moving quickly and efficiently.MANAGEMENT REVIEW SOP Template PH38 GMP, QSR & ISO Compliance
37 Best Standard Operating Procedure (SOP) Templates
GMP Protocols and Reports GMP Docs
GOOD DOCUMENTATION PRACTICES SOP Template MD25 GMP, QSR & ISO CP
Using A Standard Operating Procedure (Sop) Template In Word A Guide
GOOD DOCUMENTATION PRACTICES SOP Template PH23 GMP, QSR & ISO CP
CORRECTIVE & PREVENTIVE ACTION SOP Templates MD30 GMP, QSR & ISO
EQUIPMENT CLEANING & MAINTENANCE SOP Template PH43 GMP, QSR, ISO
STORAGE & H&LING SOP Template MD58 GMP, QSR & ISO Compliance
Q040111 DOCUMENT CHANGE CONTROL SOP GMP Templates
Easily Customized To Your Needs.
Web Standard Operating Procedure (Sop) And Accompanying Data Recording Form, Several Sample Sops, And Summaries Of The Expected Contents Of Several Types Of Sops.
A Standard Operating Procedure Is A Set Of.
Add To Cart Report Type:
Related Post: